 Having eight 3d electrons and two 4s electrons is much less energetically stable than ten 3d electrons and no 4s electrons. When d-block elements lose electrons, they lose the highest energy s electrons first, which in the case of zinc are the two 4s electrons. Follow these simple steps to find the number of protons, neutrons, and electrons for an atom of any element. For zinc, the atomic weight is 65.39, so the mass number is closest to 65. For the d-block elements, the outermost s-sublevel has higher energy than the d-sublevel, which is contrary to what the Aufbau diagram indicates. Number of Neutrons Mass Number - Number of Protons 1 - 1 0. 
Zinc is a d-block element, also known as a transition element. Electron Density Ne(cm-3):, Ion Temperature Ti(eV):, (if Ti Te). The #"Zn"^(2+)# ion has lost two electrons, which leaves it with 30 protons and 28 electrons. Electron Temperature Te(eV):, Doppler-broadened spectrum. Classified as a transition metal, Zinc is a solid at room temperature. The electron configuration of a neutral zinc atom is #"1s"^2"2s"^2"2p"^6"3s"^2"3p"^6"3d"^10"4s"^2"#. Zinc atoms have 30 electrons and the electronic shell structure is 2, 8, 18, 2 with Atomic Term Symbol (Quantum Numbers) 1S0. Zinc is a chemical element with symbol Zn and atomic number 30. 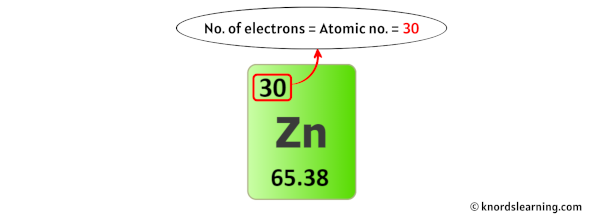
A neutral atom has equal numbers of protons and electrons, so a neutral atom of zinc would have 30 electrons. Zinc oxide is used in plastics, cosmetics, and wallpaper, while zinc sulfide is employed in luminescent dials, TV screens, and fluorescent lights.The atomic number of zinc is 30, which means that all zinc atoms have 30 protons in their nuclei. It is also used to form numerous metal alloys such as brass, bronze, and aluminum solder. In some respects zinc is chemically similar to magnesium: both elements exhibit only one normal oxidation state (+2), and the Zn2+ and Mg2+ ions are of similar size. Zinc’s principle use is to galvanize iron and steel, preventing corrosion. Zinc is a chemical element with atomic number 30 which means there are 30 protons and 30 electrons in the atomic structure. It is an essential element in the growth of plants and animals, and altered biological concentrations of zinc can lead to severe health problems. This bluish-white metal is brittle at ambient temperatures, but becomes malleable between 110–150☌. Marggraf is credited for the first isolation of zinc from the charcoal reduction of calamine in 1746. Zinc alloys have been used for centuries although, A. In the ground state A, the single bond between methylene and zinc is formed by the pairing of one of the 4spz electrons of zinc and the one electron in the sp. Use Wagner plot to help assign chemistry.Use X-ray induced Zn LMM Auger peaks, which have a larger shift with chemical state.Similarly, Zn3p (very high kinetic energy) may be observed even if Zn2p is not.If zinc is buried (under carbon, for example), Zn Auger peak may be observed even if Zn2p is not (due to difference in electron kinetic energy).Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. .jpg)
Zn2p peaks often accompanied by Auger peak at Kinetic Energy ~990eV. Zinc Transition Metal If zinc is buried (under carbon, for example), Zn Auger peak may be observed even if Zn2p is not (due to difference in electron kinetic. Element Zinc (Zn), Group 12, Atomic Number 30, d-block, Mass 65.38.For example, to write an abbreviated electron configuration for zinc atoms, we first find Zn on the. Peak width may broaden in the presence of more than one Zn species. Ionization potential: 39.722 eV Oxidation states: 2 Electrical conductivity: 0. Step 1 Find the symbol for the element on a periodic table.Zinc shows only a small binding energy shift in the Zn2p 3/2 region (1021eV - 1023eV, modal value).ZnO has an unusual O1s spectrum with two peaks, possibly indicating either Zn(OH) 2, ZnCO 3,.Zn2p peak has significantly split spin-orbit components (Δ metal=23eV).There are bigger chemical shifts observed for Zn LMM compared to Zn2p.Collect principal Zn LMM peak as well as Zn2p. The next element has two electrons and the second electron fills the 1s. 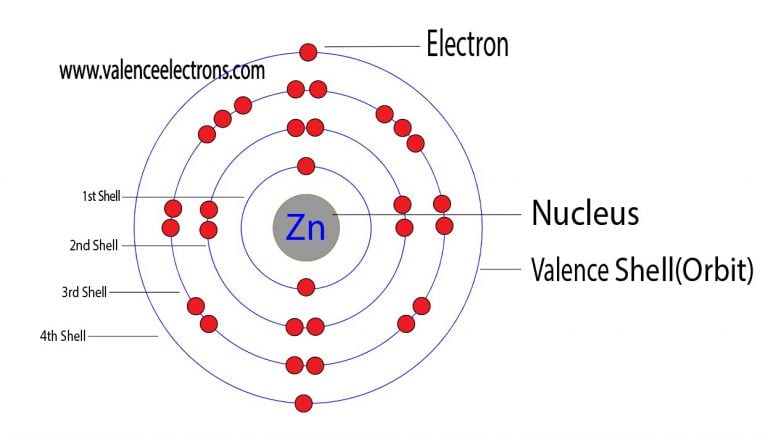
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
